I-CARE: IBD CAncer and seRious infections in Europe
I-CARE is the first observational European prospective cohort study that will provide unique information (safety, efficacy, risk-benefit ratio, and healthcare costs) on the long-term use of recommended therapy in IBD, using a predefined standardized follow-up. These real world data will be used to guide clinicians as well as Healthcare authorities to provide the best care for IBD patients by optimizing available therapies.
Recruitment began in Ireland in May 2017 and so far we have 14 active investigators who have recruited over 100 patients. We aim to recruit approximately 500 patients across 14 sites by the end of the recruitment period.
Study Design
10,206 patients with IBD will be recruited across Europe (with a 273 patients recruited in
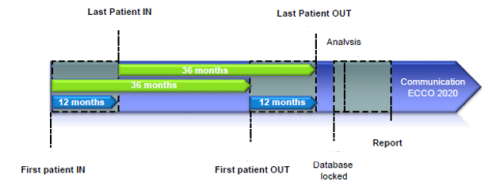
Ireland) and they were followed for three years.
Patients reported outcomes of treatment every month using a customized web based application with yearly physician validation of important events.
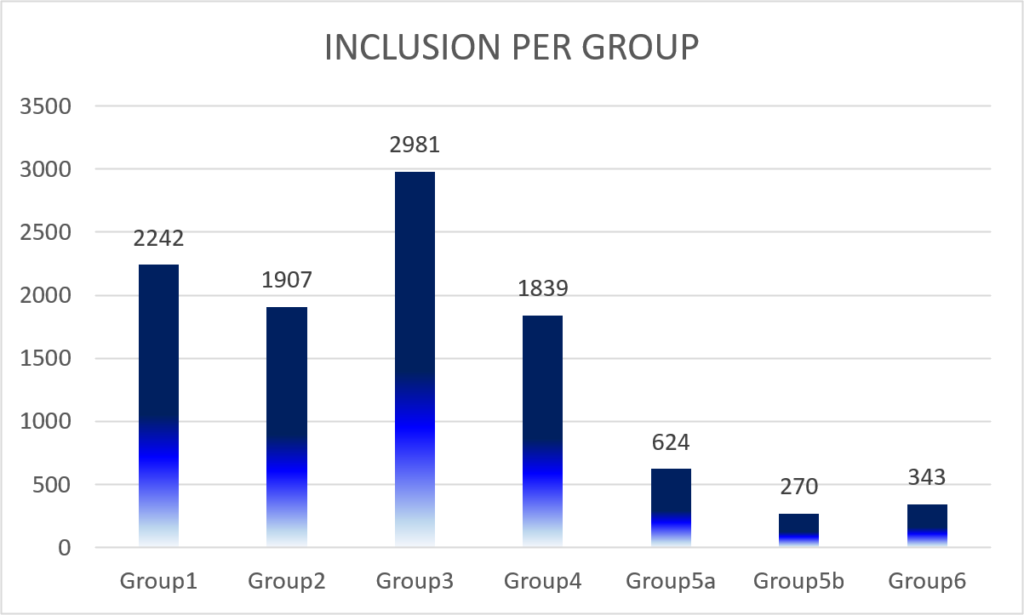
Each investigator included a total of 22 inpatients or outpatients that they personally managed for IBD, matching the inclusion criteria and stratified according to the exposure to immunosuppressive therapy at inclusion:
– Group 1: patients who have never received biological agents or immunosuppressant (all 5-ASA and steroids formulations are permitted)
– Group 2: patients receiving thiopurines alone
– Group 3: patients treated with anti-TNF therapy alone without any concomitant immunosuppressant
– Group 4: patients treated with anti-TNF therapy in combination with thiopurines or methotrexate
– Group 5a: Patients treated with vedolizumab alone (without any concomitant immunosuppressant)
– Group 5b: Patients treated with vedolizumab in combination with thiopurines or methotrexate.
– Group 6: Patients treated with ustekinumab with or without any concomitant medications.
Inclusion criteria:
- Patient with an established diagnosis of Crohn’s disease, ulcerative colitis or IBD, unclassified made at least 3 months earlier based on usual radiological, endoscopic or histological criteria.
- Patient 18 and older accepting to sign the informed participating consent form, stating that he accepts to provide personal details (mobile and home phone number, e-mail address), to complete the e-PRO as required and to be contacted by a Study Coordinator and his gastroenterologist for the purpose of the study during the entire study period and during follow up if required.
Exclusion criteria:
- Patient unable to sign the informed consent form
- Patient with no regular access to internet
- Patient refusing to sign the informed consent form
- Treatment at entry in the study with an immunomodulator different from thiopurines and methotrexate (cyclosporine, tacrolimus, mycophenolate mofetil, etc.)
- Patient previously enrolled in a Randomized Clinical Trial (If the investigational product received was blinded, and if the treatment is unknown at time of enrollment in I-CARE)
Recruitment to date
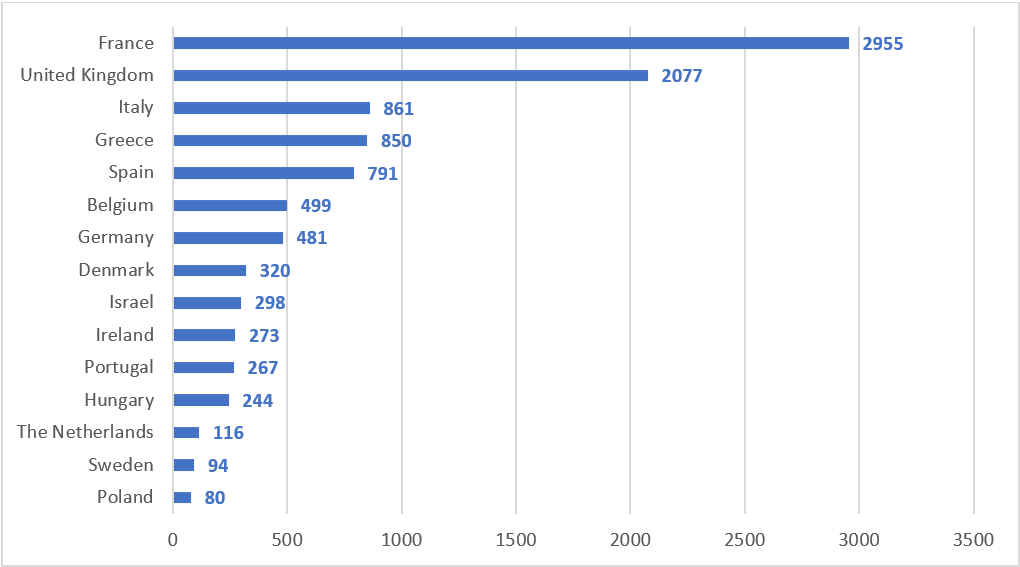
As of May 2022 I-CARE has recruited over 10,000 patients across 15 countries. Here in Ireland we have recruited 273 patients, 66% of our target. March 31st 2022 marked the end of the study with the last patient finishing the study on the 16th of May 2022.
I-CARE included 508 principal investigators, 305 study nurses, and 22 contributors across the 15 countries included.
Results
A total of 10,206 patients were enrolled between March 2016 and April 2019, including 6169 (60.4%) patients with Crohn’s disease, 3853 (37.8%) with ulcerative colitis, and 184 (1.8%) with a diagnosis of IBD unclassified. Thirty-two percent of patients were receiving azathioprine/thiopurines, 4.6% 6-mercaptopurine, and 3.2% methotrexate at study entry. At inclusion, 47.3% of patients were treated with an anti-tumor necrosis factor agent, 8.8% with vedolizumab, and 3.4% with ustekinumab. Roughly one-quarter of patients (26.8%) underwent prior IBD-related surgery. Sixty-six percent of patients had been previously treated with systemic steroids. Three percent of patients had a medical history of cancer prior to inclusion and 1.1% had a history of colonic, esophageal, or uterine cervix high-grade dysplasia.
DOI: 10.1016/j.cgh.2022.09.018