MOBIDIC: Molecular Biomarkers and Adherent and Invasive Escherichia Coli (AIEC) Detection study In Crohn’s disease patients
Abnormal colonization of the gastrointestinal tract by Enterobacteriaceae might play a role in the development of Crohn’s Disease (CD). Clearance of this bacteria could represent a new therapeutic strategy in CD. The purpose of this study was to develop a non-invasive test to look for a specific bacteria, invasive Escherichia Coli, (AIEC) known to be found in certain patient’s with Crohn’s Disease. High levels of AIEC have been associated with premature recurrence of acute flare ups of the disease, longer lasting flare ups and more cases needing surgery.
Enterome Bioscience, the company sponsoring the study, are aiming to develop a method of detecting AIEC in stool samples so that an invasive procedure, like a colonoscopy, may not be necessary. This may allow AIEC to be identified in some patients thought to be at risk and enable treatments targeted to the bacteria.
This study enrolled 143 in Europe (75%) and in the USA (25%). Of those, 111 completed the study. The results showed 61% of CD patients mainly in remission were colonized by this specific bacteria. A significant association between disease activity and stool analysis were established which will allow Enterome Bioscience to pursue the development of this method in future clinicals trials with EB8018, a bacterial protein inhibitor.
Results
We analyzed the metagenomic composition of the gut microbiome of 358 patients with CD from two different cohorts and characterized the presence of FimH-expressing bacteria. To assess the pathogenic role of FimH, we used human intestinal explants and tested a specific FimH blocker to prevent bacterial adhesion and associated inflammation. We observed a significant and disease activity-dependent enrichment of Enterobacteriaceae in the gut microbiome of patients with CD. Bacterial FimH expression was functionally confirmed in ileal biopsies from 65% of the patients with CD. Using human intestinal explants, we further show that FimH is essential for adhesion and to trigger inflammation. Finally, a specific FimH-blocker, TAK-018, inhibits bacterial adhesion to the intestinal epithelium and prevents inflammation, thus preserving mucosal integrity.
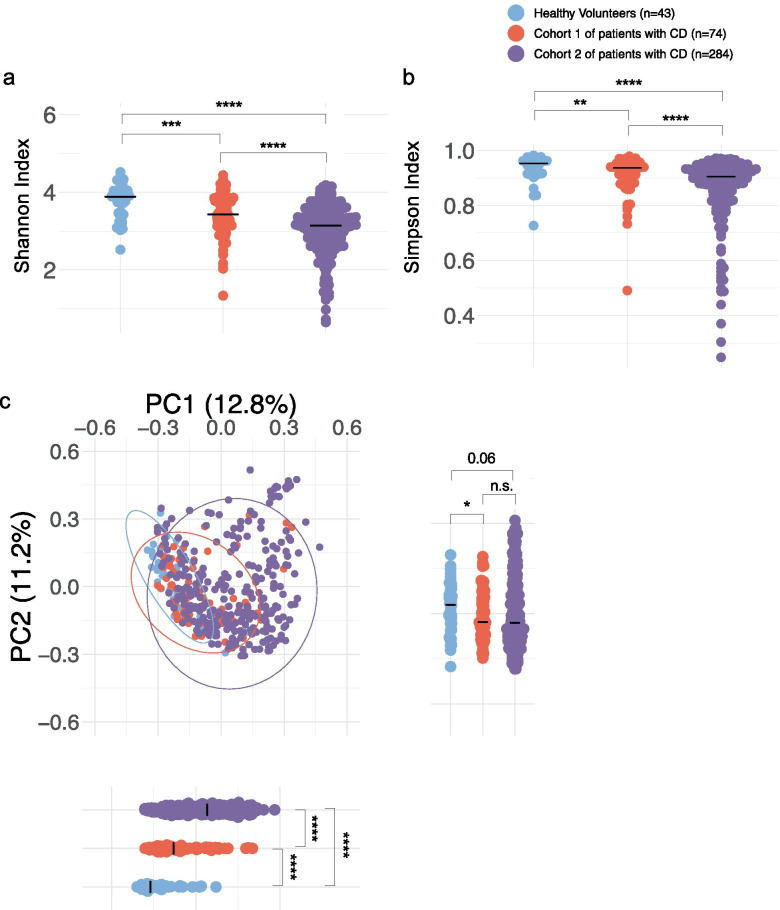
Altered microbiota profiles in patients with CD from 2 different cohorts. a, b Species diversity in patients with CD and healthy volunteers were calculated using alpha diversity: Shannon (a) and Simpson indices (b). c Microbial clustering is shown based on Bray–Curtis dissimilarity principal coordinate analysis (PCoA) metrics for Cohort 1, Cohort 2, and HV.
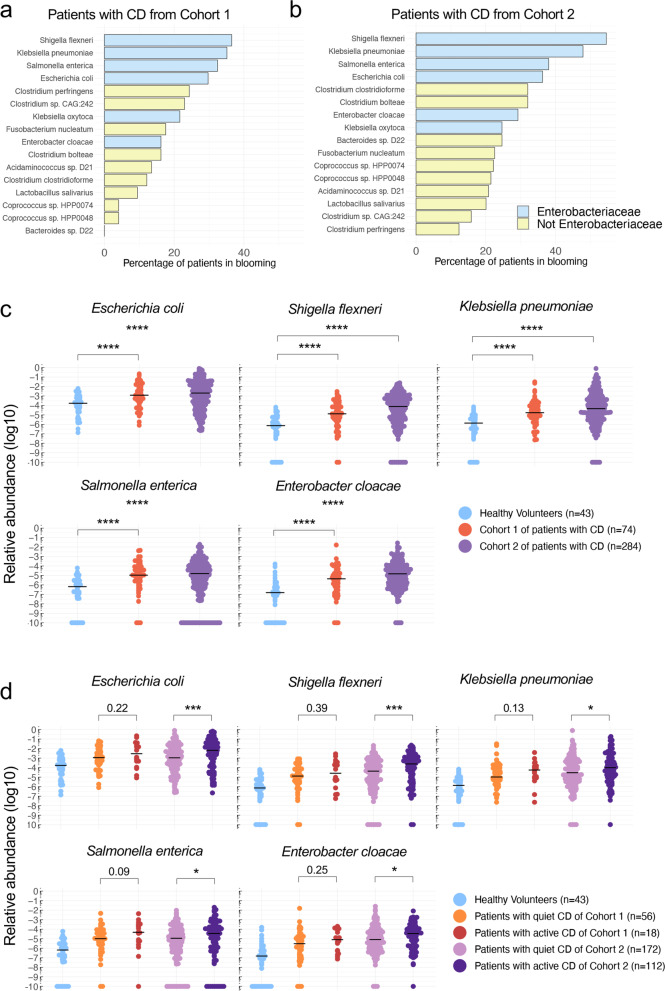
Fecal microbial composition of patients with CD is characterized by a blooming of Enterobacteriaceae. a, b Top blooming species by percentage of patients with CD in Cohort 1 (a) and Cohort 2 (b), for whom a tenfold increase has been observed compared with mean relative abundance in HV. Are represented only the species with at least 20% of patients in blooming either in Cohort 1 or in Cohort 2. Note the presence of 4 Enterobacteriaceae among the top 5 species. c Relative abundance of Enterobacteriaceae spp in HV and patients with CD from Cohort 1 and Cohort 2. d Relative abundance of Enterobacteriaceae spp in HV, active and quiet patients with CD from Cohort 1 and Cohort 2.
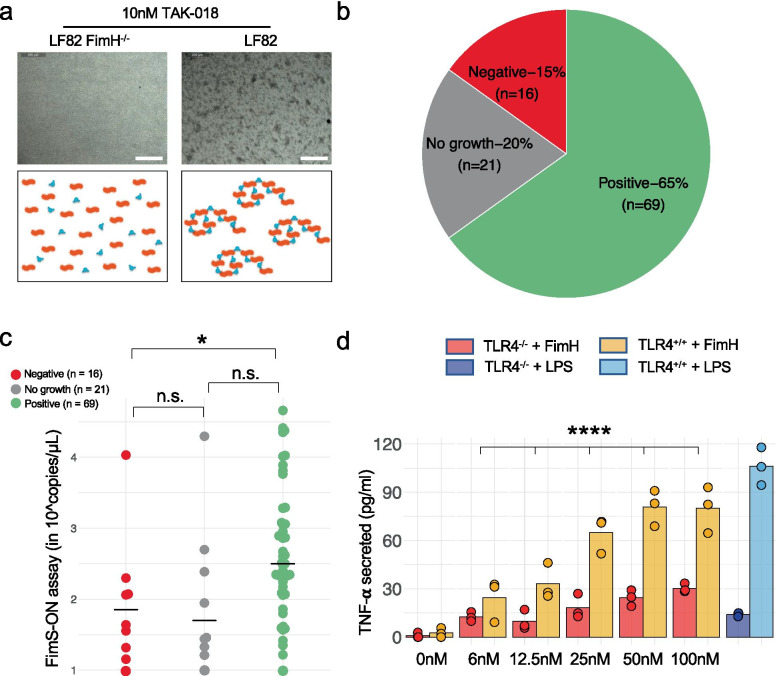
FimH adhesin is expressed by bacteria in ileal biopsies from patients with CD and induces inflammatory response in a TLR4-dependent manner. a Aggregation of LF82 E. coli upon incubation with TAK-018. Note that FimH−/− LF82 E. coli did not aggregate upon incubation with E. coli illustrating that aggregation is a FimH-dependent phenomenon. b Presence of FimH-expressing bacteria in patients with CD as measured by aggregation of bacteria to TAK-018 in ileal biopsies. One to two biopsies were analyzed per patient. c FimS-ON expression in fecal samples from patients with CD, as measured by qPCR. Bars represent the median of all points. Nonparametric Mann–Whitney U test was used to identify the statistically significant differences between groups (*P < 0.05; n.s. nonsignificant). d Secretion of TNF-α of HEK-hTLR4+/+ and HEK-TLR4−/− cells upon incubation with increased concentration of FimH. LPS was used as a positive control at a concentration of 100 ng/ml. FimH was produced in yeast in order to be LPS-free.
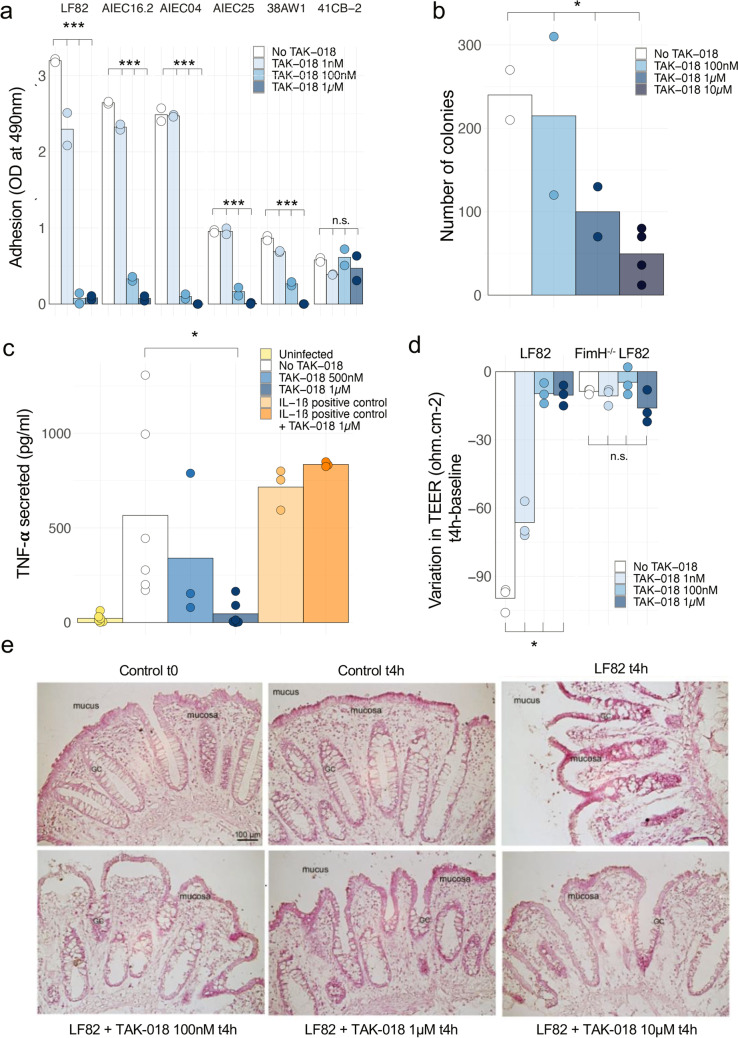
FimH blocker TAK-018 prevents Enterobacteriaceae adhesion and inflammation of gut explant. a Adhesion of different strains of E. coli to human primary intestinal epithelial cells, in the presence of increasing concentrations of TAK-018. Note that the 41CB2 strain did not express FimH. Bars represent the mean value of individual points which themselves correspond to biological replicates. ANOVA was performed for each group to identify the statistically significant effect of TAK-018 (***P < 0.001). b Number of intracellular colonies in epithelial cells from human ileal explant incubated with LF82 E. coli, in the presence of increasing concentrations of TAK-018. Bars represent the mean value of individual points which themselves correspond to biological replicates. ANOVA was performed to identify the statistically significant effect of TAK-018 (*P < 0.05). c TNF-α secretion of human ileal explant incubated for 4 h with 109 LF82 E. coli, in the presence of increasing concentrations of TAK-018. IL-1β was used as a positive control to trigger inflammation. Bars represent the mean value of individual points which themselves correspond to biological replicates. A nonparametric Wilcoxon signed-rank paired test was used to identify the statistically significant differences between the LF82 and LF82 + TAK-018 1 µM groups (P = 0.031). A linear mixed model was used to identify the statistically significant differences between the groups LF82, LF82 + 500 nM, and LF82 + 1 µM (P = 0.07). d Variation of transepithelial electric resistance (TEER) of T84 cells incubated for 4 h with 109 LF82 E. coli, in the presence of increasing concentrations of TAK-018. Note that FimH−/− LF82 E. coli does not adhere to T84 cells and therefore does not affect TEER. Bars represent the mean value of individual points which themselves correspond to biological replicates. A nonparametric Kruskal–Wallis test was used to identify the statistically significant differences between the LF82 groups (P = 0.02). e Histopathologic effects of LF82 E. coli adhesion on human ileal explants from a patient with CD and respective countereffects in the presence of increasing concentrations of TAK-018.
Conclusion
We propose that TAK-018, which is safe and well tolerated in humans, is a promising candidate for the treatment of CD and in particular in preventing its recurrence.
To access the paper, click HERE