SECURE-IBD: Surveillance Epidemiology of Coronavirus Under Research Exclusion in Inflammatory Bowel Disease
SECURE-IBD is an international, pediatric and adult database to monitor and report on outcomes of COVID-19 occurring in IBD patients.
With the collaboration of our entire IBD community, we will rapidly be able to define the impact of COVID-19 on patients with IBD and how factors such as age, comorbidities, and IBD treatments impact COVID outcomes. This database was active from March 2020 to January 2022.
Key points:
- This was an international effort—we are counting on robust participation and collaboration.
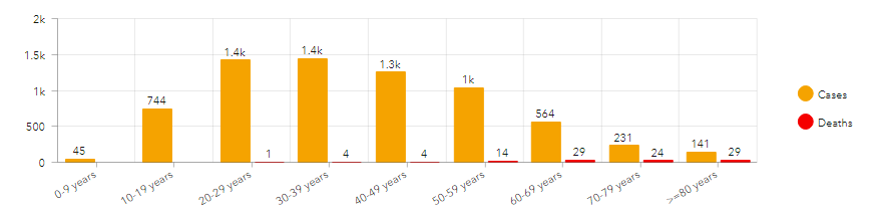
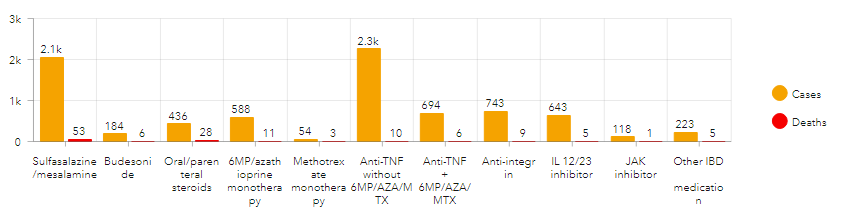
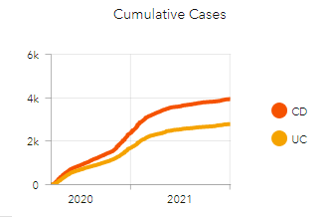
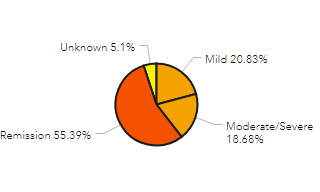
- We provided the IBD community with regularly updated summary information about reported cases, including numbers of cases by country, number of cases by treatment, etc. so the entire IBD community has access to these data.
- The database contains only de-identified data, in accordance with HIPAA Safe Harbor De-Identification standards.
- The UNC-Chapel Hill Office for Human Research Ethics has determined that storage and analysis of de-identified data does not constitute human subjects research as defined under federal regulations [45 CFR 46.102 and 21 CFR 56.102] and does not require IRB approval.
Findings:
Through SECURE-IBD, we have learned that age and increased number of comorbidities are associated with adverse COVID-19 outcomes among patients with IBD, and that Hispanic and Black individuals with IBD tend to have worse COVID-19 outcomes than white individuals. We found that corticosteroids are associated with more severe COVID-19 outcomes, while biologic medications are not. Overall, our data have reassured the international IBD community that patients with IBD can continue their medications during the pandemic, with the caveat of weaning steroids when possible.
We have also learned many valuable lessons that can be applied to future projects requiring rapid data collection during a crisis. Engaging the IBD community early in the process and working with international organizations/sponsors led to a level of participation that we never dreamed possible. We remain astounded at the unwavering commitment of our community to come together to answer critically important questions for patients with IBD. Adaptability played another key role in our success; Adding new questions to our case report forms in response to emerging clinical needs and re-evaluating initial conclusions based on new data enabled us to keep pace during a rapidly changing situation.
As we move into a new phase of the pandemic with different variants, evolving vaccines and vaccination strategies, re-infections, and COVID-specific treatments, the most pressing questions have shifted away from “What are the medication and other risk factors for developing a more severe course of COVID-19 in patients with IBD?” to new questions about vaccines, immunity, testing, and treatments.
To access the interactive map of the database, click HERE