STEIG: Stelara Treatment Effectiveness in Irish Gastroenterology
STEIG is a retrospective study of clinical outcomes of off licence treatment with Ustekinumab for Crohn’s Disease. Ustekinumab (Stelara) is a monoclonal antibody against the common p40 subunit of IL-12 and IL-23 and has recently been approved for (intravenous) induction and (subcutaneous) maintenance treatment of patients with moderately-to-severely active Crohn’s disease (CD). However subcutaneous USK has been used off label for several years in patients refractory to approved therapies. We aimed to evaluate the real-world effectiveness of subcutaneous USK in medium-to-long term CD treatment outcomes.
Data Collection
Data was collected on:
- age
- gender
- disease location and phenotype
- disease duration
- smoking status
- prior and concurrent therapies
- body weight/BMI
- Ustekinumab induction and maintenance dosing
- duration of treatment
- need for hospitalisation and/or surgery during follow-up
End-Points
The primary end-point will be treatment success, defined as maintenance therapy without need for hospitalisation due to disease flare or for surgical resection.
The secondary end-points will focus on loss of response and treatment related adverse events.
Multi-variate analysis of clinical factors determining treatment success and time to treatment failure will be performed.
Methods
A multicentre retrospective study, entitled STEIG (Stelara Treatment Effectiveness in Irish Gastroenterology; which is the Irish language translation of ‘intestine’) was performed through the INITIative IBD research network (www.initiativeibd.ie). Patients with confirmed Crohn’s Disease (CD) who were treated off label with USK between 2011 and 2016 were identified. Demographic data, disease characteristics, disease duration, surgical history, medication history e.g. anti-TNFα and Immunomodulator exposure, endoscopic and biochemical data was collated. In addition, induction dosing regimens and details of dose optimisation from the 6 participating centres were examined.
The outcome measures which were assessed included:
• Dose and response to SC induction dosing
• Maintenance strategies
• Sustained benefit at 12 & 24 months defined as treatment continuation and clinical benefit from USK, as determined by treating physician, without need for surgical resection
• Changes in Endoscopic/Biochemical parameters.
• Surgical intervention during follow-up period
• Time to surgery/treatment discontinuation
Results
62 patients from 6 centres were included. Median age was 39 years (IQR 32.75-49yrs) and median disease duration was 14 years (IQR 9.75-23.25yrs). All patients were refractory to anti-TNF therapy. 60 patients responded to induction therapy with subcutaneous USK and proceeded to maintenance therapy. Mean cumulative induction dose was 220mg (SD 40mgs), given exclusively by subcutaneous injection. Standard maintenance dose was 90mg 8-weekly with 21 patients requiring dose escalation during median follow up of 20 months. 64% had sustained benefit at 12 months and >80% of these patients continued to benefit at 24 months. 42% of patients demonstrated endoscopic improvement with USK therapy. 11/62 patients (18%) required surgical intervention within 12 months of starting therapy.
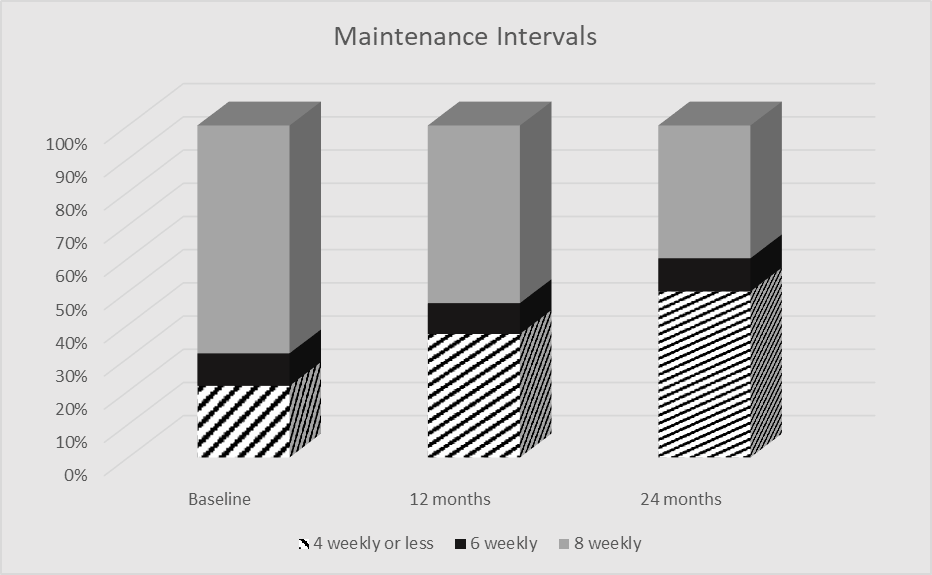
Proportion of patients treated with 90mg USK at ≤4, 6 or 8 weekly intervals at baseline, 12 and 24 months.
Authorship
Investigators across six sites included Catherine R. Rowan & Alaa Alakkari, Carthage Moran, Jim O’Connell, Garret Cullen, Hugh E. Mulcahy, Aoibhlinn O’Toole, Juliette Sheridan, Jan Leyden, Padraic MacMathuna, Susan McKiernan, David Kevans, Colm O’Morain, Deirdre McNamara, Glen A. Doherty & Barbara M. Ryan.