VARIATION: Reduced serological response to COVID-19 vaccines in patients with IBD is further diminished by TNF inhibitor therapy
VARIATION was a prospective multi-centre Irish study aiming to determine the impact of IBD and various medications for treatment of IBD on antibody responses to vaccination against COVID-19.
Background and aims:
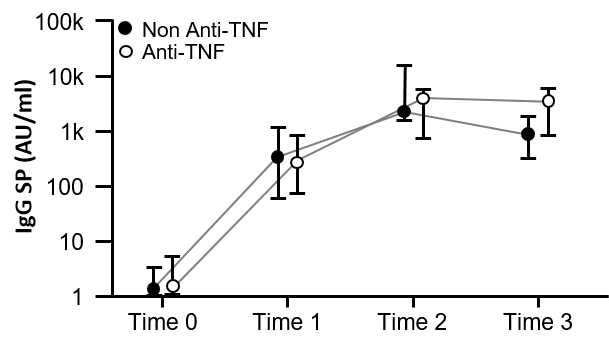
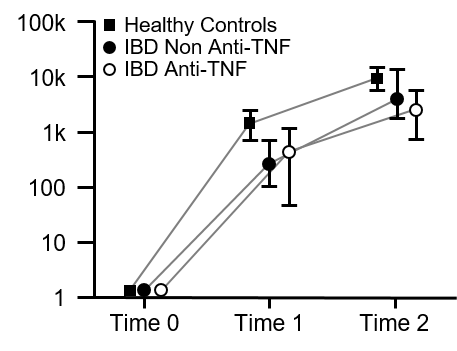
Evidence suggests patients with inflammatory bowel disease [IBD] receiving TNF antagonists have attenuated response to vaccination against COVID-19. We sought to determine the impact of IBD and of various medications for treatment of IBD on antibody responses to vaccination against COVID-19.
Methods:
Patients with IBD (n=270) and healthy controls (n=116) were recruited prospectively and quantitative antibody responses assessed following COVID-19 vaccination. The impact of IBD and medications for treatment of IBD on vaccine response rates was investigated.
Results:
Patients with IBD have attenuated serological responses to SARS-CoV-2 vaccination. Use of anti-TNF therapy negatively affects anti-SP IgG levels further. Patients who do not seroconvert following vaccination are a particularly vulnerable cohort. Impaired responses to vaccination in our study highlight the importance of booster vaccination programmes for patients with IBD.